At Arterius we support global healthcare innovators to develop next-generation, patient-centric products. Our current focus is on improving medical devices for the treatment of peripheral arterial disease
Technology foundation
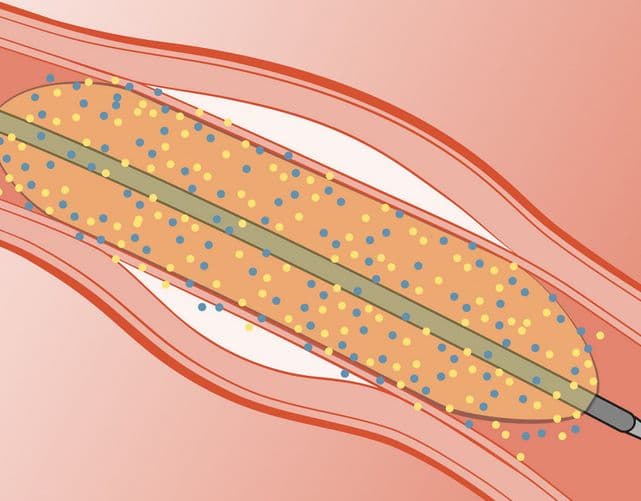
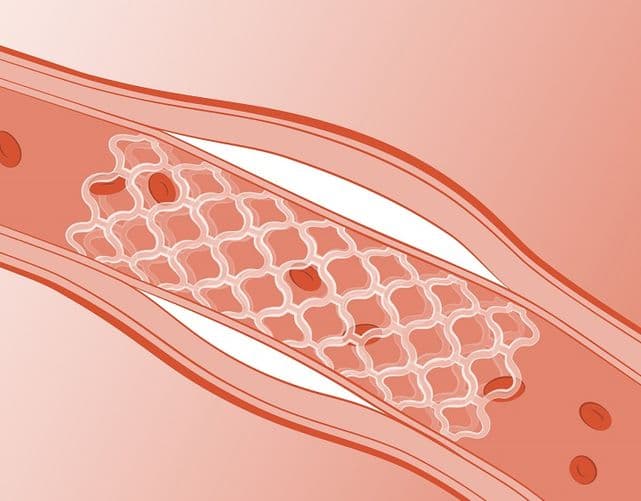
Arterius has developed two advanced polymer platforms for use in devices to manage arterial disease while leaving no long-term implants in the body


Arterius has raised over
£10 million
in grant and equity funding
to bring its two novel technology platforms to proof of concept
Partners
Arterius has collaborated with a range of esteemed academic, clinical and industry partners during the development of its core technologies.

Innovate UK

University of Southampton

University of Bradford

University of Nottingham

University of Bristol

Imperial College






-1-640x718.png&w=1920&q=75)

